Welcome to week six of the blogging challenge. This week is all about sciencey scientific science stuff. Say that three times fast. No, actually. I’m dead serious, say it three times fast. So for this week we got to choose what science thing we actually wanted to learn a bit about. I’ve decided to learn about the periodic table of elements and how atoms and ions mix together. I’ve decided to do this post on that because of two things. One, I find it very interesting, and two, we are already learning about this in Scimatix so this post should help me get a little ahead and better understand this unit.
The periodic table is above. If you don’t already know, the periodic table is all the elements placed in order of their atomic number. Today we are going to learn about how the elements bond. Let’s take an element with – 2 net charge. It would bond well with an element of + 2 net charge. This would then make a compound. The net charged is based on how many protons and neutrons you have compared to electrons. If you have more electrons it would be negative, and if you have more protons or neutrons it would be positive. There is a great site that you can simulate creating an element. Go here to see it.
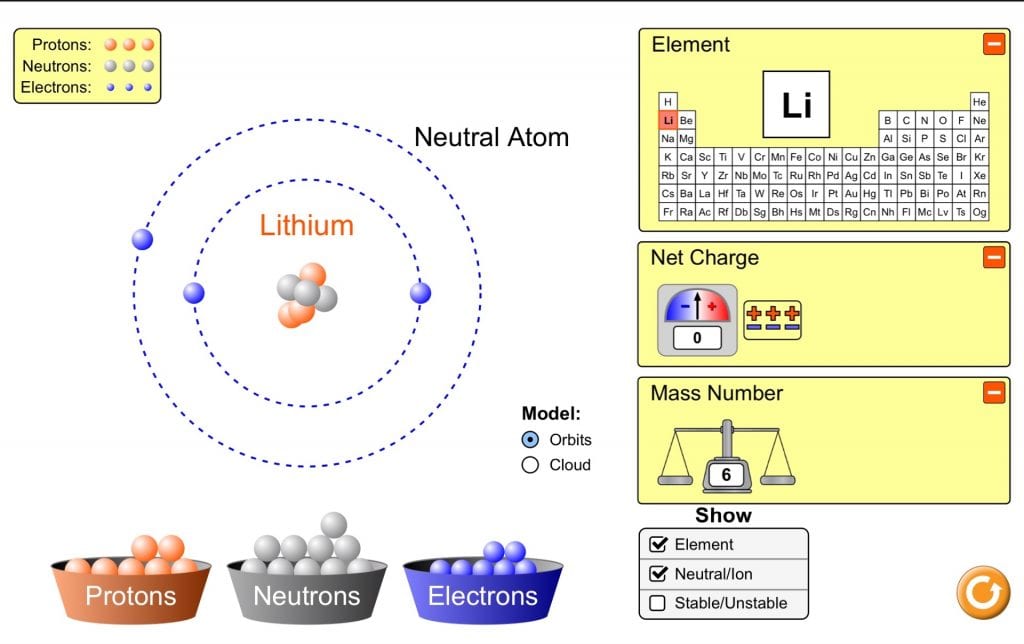
Above you can see I created Lithium. There are the same amount of protons as electrons so the net charge is 0.
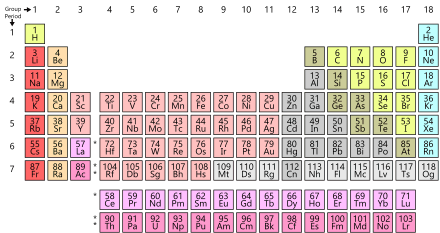
November 16, 2018 at 6:55 pm
Lit fire my dude
November 16, 2018 at 6:56 pm
Holy moly this is absolute gold. Beutifully written, creative title, and great pictures. You the real deal my dude.
November 16, 2018 at 7:05 pm
This is so good. I can really appreciate all the attention to detail. Great job.
November 16, 2018 at 7:08 pm
OMG!!! This is AMAZING. HOLY! OMG!
November 16, 2018 at 7:09 pm
This is one of the best posts I’ve seen in a while. The picturesque beauty of it all is absolutely exquisite. The brilliancy of the title is godlike. The creativity is beyond unique. Keep up the great work.
Sincerely, Luciano
November 16, 2018 at 7:12 pm
Holly this is a great post I am amazed how you completed it so quickly and added some many pictures, videos and links. Great job